Project Lead
Associate Professor Marie-Liesse Asselin-Labat
Full bio
COLLABORATIVE NETWORK
MACH Omics
Learn moreLead partner
WEHI
MACH PARTNERS
Royal Melbourne Hospital
Peter Doherty Institute for Infection and Immunity
From the onset of tumour growth, tumour and immune cells evolve together, where each cell sends signals to other cells present around them, influencing their behaviours. On the one hand, tumour cells may develop mechanisms to become invisible to the immune system. On the other hand, immune cells may lose their capacity to eliminate cancer cells effectively.
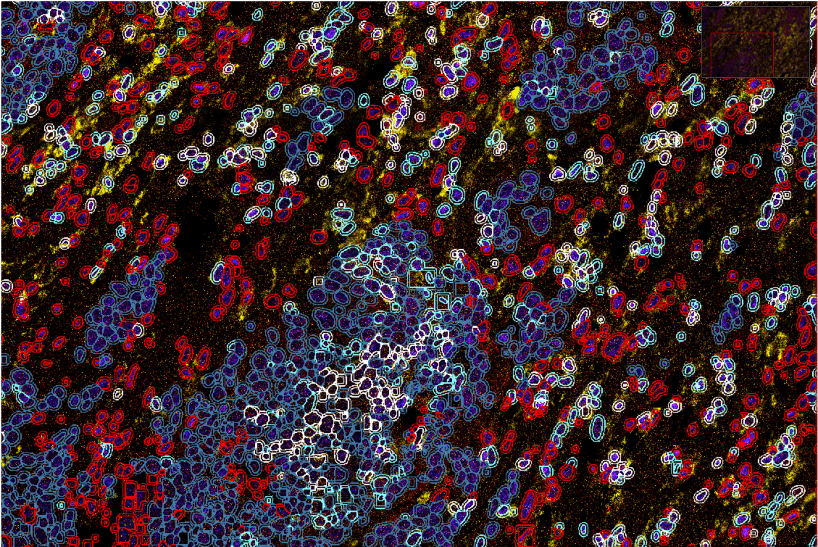
We aim to explore the impact of immune cells on tumour cell evolution and vice versa from the perspective of each cell type. Our team uses cutting edge technologies to characterise each cell and its relationship with neighbouring cells in human cancer biopsies. Our unique approach integrates molecular cell characteristics and spatial cell organisation using algorithms based on artificial intelligence to determine the state in which a cell resides and how cells transit between different states under the influence of surrounding cells.
Our project takes a spatial multi-omic approach combining spatial proteomic, transcriptomics and genomics analyses of non-small cell lung cancer samples to understand how the immune microenvironment drives tumour evolution.
- Understanding lung cancer evolution
- Identifying mechanisms of resistance to immunotherapy
- Identify novel therapeutic targets
- Determine risk of recurrence
Weeden, C. E., Gayevskiy, V., Trussart, M., Marceaux, C., Tubau Ribera, N., Batey, D., Teh, C. E., Mitchell, A. J., Antippa, P., Leong, T., Steinfort, D., Irving, L., Gordon, C. L., Swanton, C., Speed, T. P., Gray, D. H., Asselin-Labat, M.-L. Early immune pressure imposed by tissue resident memory T cells sculpts tumour evolution in non-small cell lung cancer. bioRxiv 1–46 (2021). doi: 10.1101/2021.04.20.440373.